|
We write \(S^o_A\left(T\right)\) to indicate the absolute entropy of substance \(A\) in its standard state at temperature \(T\).\) illustrates this procedure for the combustion of the liquid hydrocarbon isooctane (C 8H 18 2,2,4-trimethylpentane). It is usually included in compilations of thermodynamic data for chemical substances. The standard entropy is usually given the symbol \(S^o\). Absolute entropy is the total amount of entropy needed to heat an object from absolute zero (where entropy and temperature are equal to zero) to the desired. Although entropies and heat capacities are thermodynamic features encoded rather globally in the shape of the PES,25,26 conformations can be used to map the problem to well-de ned points on the PES. This formula defines the entropy up to an arbitrary constant S0. 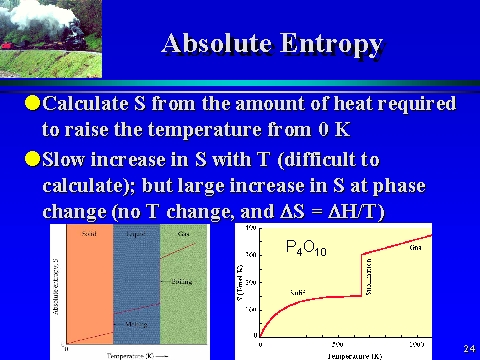
When the entropy value is calculated for one mole of the substance in its standard state, the resulting absolute entropy is called the standard entropy. Temperature derivatives of the entropy are related to heat capacities: (S. Suppose, it is required to determine absolute entropy. Where the substance undergoes phase changes, the contribution that the phase change makes to the entropy of the substance is equal to the enthalpy change for the phase change divided by the temperature at which it occurs.Īt any given temperature, the entropy value that is obtained in this way is called the substance’s absolute entropy or its third-law entropy. Combining the heat capacity data with the enthalpy data on phase transformations, the absolute entropy. marks the transition from the low- to high-temperature phase of quartz and is the melting temperature. To determine the absolute entropy of a substance at a given temperature, one can use standard entropies of substances at 25C and then apply the heat capacity. Phase changes are isothermal and reversible. Molar entropy,, and enthalpy,, of quartz at constant pressure,, as a function of absolute temperature,, by integrating the molar specific heat capacity and entropy capacity of Figure 3. 
Determine the heat capacity of 3000 J of heat is used to heat the iron rod of mass 10 Kg from 20oC to 40oC. Assigning a value of zero to S0 allows the absolute value of the. 5.4 Dimensionless heat capacity 5.5 Heat capacity at absolute zero 5.6 Solid phase 5.7 Theoretical estimation 5.8 Calculation from first principles 5.9. In temperature ranges where experimental heat capacity data are available, the entropy change is obtained by integration using these data. Temperature difference T 20 o C, Heat lost Q 300 J. In this lecture, we examine the heat capacity as a function of temperature, compute the. The standard molar entropy of Al2O3 Al 2 O 3 is obtained by adding the Debye. \), using Debye’s theoretical relationship, \(C_P=AT^3\) \(A\) is obtained from the value of \(C_P\) at the lowest temperature for which an experimental value of \(C_P\) is available. Once you have obtained a reasonable fit to the heat capacity data, calculate the ‘non-Debye’ contribution to the standard molar entropy by dividing your polynomial function, P(T) P ( T), by T T and integrating the result from the low temperature limit up to 298 K.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
